"Planting green wires+new energy+energy storage" to ensure the stability of power supply to the grid.
CCTV News:On June 11th, China’s first UHV project to send electricity outside the wind power photovoltaic base in desert, Gobi and desert areas — — Construction of 800 kV HVDC transmission project from Ningxia to Hunan started.

The total investment scale of Ningdian’s project in Hunan is about 100 billion yuan, starting from Zhongwei in Ningxia and passing through Gansu, Shaanxi, Chongqing, Hubei, Hunan and other five provinces (regions) and one city, with a total length of 1,634 kilometers. The project mainly transports green electricity and clean electricity produced by using clean energy such as solar energy and wind energy in desert, Gobi and desert areas, and has become an innovative project in Ningxia to achieve the goal of "double carbon" and a demonstration project to build a new power system and realize the optimal allocation of new energy in a large scale.

It is understood that the total installed capacity of supporting power supply for Ningdian’s project in Hunan is 17.64 million kilowatts, which will promote the transmission capacity of Ningxia power grid from 14 million kilowatts to 22 million kilowatts, and the installed capacity of new energy will exceed 50 million kilowatts, which will greatly promote the local consumption of new energy in Ningxia and optimize the allocation of new energy on a large scale, and fundamentally solve the problem of grid-connected consumption of new energy in central and southern Ningxia. After the project is put into production, it will increase Hunan by 36 billion kwh every year — 40 billion kwh of electricity consumption.
Ecological management of "planting green and protecting wires" makes the electric tower stronger
Ningdian’s project into Hunan is the first large power channel in China, which mainly focuses on developing desert photovoltaics and transporting new energy. What difficulties did the owner encounter in the pre-planning and current construction process? How is it solved?

The total length of Ningdian project in Hunan is 1634 kilometers, and the Ningxia section is 261 kilometers. Among them, 68% of the lines pass through desert, Gobi and desert terrain, which means that some power towers need to be built in the "Shage Desert" area, and the sparse vegetation and complex terrain in the area, coupled with the invasion of sandstorms, will make the power towers fall down, threatening the safe and stable operation of transmission lines.

In order to solve this problem, the project owner used the previous experience of UHV operation and maintenance management for reference, and launched the "Green Line Protection" plan.
"New energy+energy storage" ensures the stability of power supply to the network.
In addition, the engineering construction team also explored the technology of "new energy+energy storage" to solve the technical problem of unstable new energy.

In order to master the law of new energy power generation, Ningxia electric power department has stepped up scientific and technological research, developed and established an early warning system, and used artificial intelligence to process and analyze massive data to monitor the operation of new energy power in real time.

At the same time, in order to ensure the stable transmission of new energy power, Ningxia electric power system continues to tackle key problems of "new energy+energy storage" technology, providing strong support for project construction.
When the Director of the Cultural Tourism Bureau changes to online celebrity, can the "flow" change to "reservation"?
Guangming Daily reporter Zhang Zheng Guangming Daily correspondent Zhang Ziqi Deng Bailu
Xie Wei, director of the Cultural Tourism Bureau of online celebrity, Suizhou City, Hubei Province, who was both "spitted" and happy by netizens because of her hair accessories and slightly fat figure, told reporters with joy: "In the first quarter of this year, the city received a total of 7.526 million tourists, achieving a tourism income of 4.67 billion yuan, exceeding the data of the same period in 2019 and setting a record high."
Contrary to the image of the video, he is meticulous at work. Recently, when he spot-checked a problem at a scenic spot, he seriously told the staff of the local cultural tourism system: "If the traffic goes up and the word-of-mouth drops, we will only be bitten by the traffic!"
In the past two years, the directors of cultural tourism bureaus in various places have been "very busy": they have to "show their faces" in a high-profile way to promote cultural tourism, and they have to bear the pressure brought by the network — — Some people call it "the director trapped in traffic."
In the face of some controversy and traffic, Xie Wei is optimistic, "willing to appear in the camera ‘ Volume ’ The director of the cultural tourism bureau still hopes to do something. " After all, he did not forget to tell reporters to give more publicity to Suizhou.
Many readers are curious, why did they become online celebrity? What kind of confusion are you facing? Can you maintain the heat for a long time? To this end, Guangming Daily reporter conducted an in-depth interview with this group.

Tourists visit Hongcun Scenic Spot in yi county, Huangshan City, Anhui Province. Photo by Xu Jiadong/Bright Picture
Why did they become online celebrity?
No matter in appearance or expression, Xie Wei seems to have nothing to do with the standard of "online celebrity". In the video that made him out of the circle, Xie Wei wore Hanfu, which quickly attracted ridicule from netizens. "I knew from the beginning that I was very sloppy, but at that time, I felt that this would not affect the beauty of the Millennium Ginkgo Valley. I didn’t expect netizens to focus on me. " Xie Wei said.
Being online celebrity is not Xie Wei’s initial intention. In February 2022, Xie Wei was transferred from Suixian County to Suizhou City, Hubei Province as the director of the Culture and Tourism Bureau. During the epidemic prevention and control period, the local cultural tourism industry suffered heavy losses. Xie Wei took office for a month, which coincided with the peach blossom season in Suizhou. He confidently invited more than 20 people from the media to promote it. Unexpectedly, after the video was sent out, the response was flat and the publicity effect was not satisfactory.
Xie Wei called "meat pain" after calculating the account. "An activity, each big V needs hundreds to thousands of dollars in labor costs. If there are such activities every month, the annual budget will be several hundred thousand smaller. Comparing the effect, spending too much money is not a long-term solution. "
At that time, Liu Hong, director of the Cultural Tourism Bureau of Ganzi Prefecture, Sichuan Province, had become popular on the Internet with his martial arts costume and became one of the first directors of online celebrity, attracting a lot of attention.
This made Xie Wei see new possibilities. "I thought, why not follow the example of Director Liu Hong and try it yourself? It will save more money than inviting stars and big V." Xie Wei said.
Whether there is controversy or not, we have to admit that Xie Wei’s "explosion" has indeed saved a lot of money for Suizhou’s image promotion.

Tourists visit the drum culture exhibition at the Bell and Drum Tower Museum in Xi ‘an, Shaanxi. Xinhua news agency
Xie Wei, who took off the label of "online celebrity", is the most typical image of a civil servant in the public mind: wearing a dark coat with a white shirt, showing a calm temperament in his gestures. Before entering the cultural tourism system, he served as secretary of the township party Committee and participated in flood control and disaster relief work.
In his view, high-profile popularity is not contrary to his original intention. What the tourism industry needs is "marketing like a madman". Xie Wei said frankly, "online celebrity officials are just a statement. Actually ‘ Red ’ It’s a flash, and civil servants are our job. I can’t accept myself when everyone is writing ‘ Do not move ’ This will be sorry for the trust of the organization. "
Ouyang Min, an associate professor at the School of Journalism and Communication, Wuhan University, believes that the change from "low-key" in grass-roots work to "high-key" in cultural tourism marketing actually reflects the functional transformation of government departments from "managers" to "servers".
"For a long time, the image of officials has always been serious. In this upsurge, the directors took the initiative to ‘ Decapitation ’ In front of the camera, the expression of escaping and youthfulness subverts the public’s inherent cognition of civil servants. The interweaving of online celebrity and officials has produced a strong sense of comedy and conflict, which accords with the audience’s ‘ Curious ’ Psychology. " Ouyang Min believes that this is a development opportunity for the local cultural tourism industry.
What are the dilemmas in traffic?
The directors at the center of the topic found that traffic brought not only economic benefits, but also many controversies and rumors.
"I don’t know if you have seen the video circulating on the Internet? In their video, I was turned into a little girl in her twenties, wearing a sling and ogling everywhere. " When talking about this matter, Xie Wei frowned and looked very helpless.
In March of this year, some self-media AI-changed Xie Wei’s video and marked it with the words "Xie Director’s modern video footage" in an attempt to share a piece of the traffic tide, which also misled many netizens.
After the video was released, negative comments on the Internet flooded into Xie Wei. Someone asked him in the comment area: "Is it necessary to sell ugliness because of ugliness?"
Submerged in the voice of public opinion, Xie Wei said frankly that he once felt at a loss. He is afraid that he will be labeled as "grandstanding" and even more worried that the surging bad reviews will affect the image of Suizhou. "Because of this incident, many fans have withdrawn from my fan base, and the video click rate has also been affected." Xie Wei said.

In Bainiu Village, Changhua Town, Lin ‘an District, Hangzhou, Zhejiang Province, Luo Xingyue (first from right), an investment operator of the village scenic spot, introduced the natural scenery of Bainiu Village to tourists. Xinhua news agency
While Xie Wei is facing confusion, directors in other places have also begun to examine the "double-edged sword" of traffic.
In March last year, director of the Cultural and Tourism Bureau of daofu county, Sichuan Province, Jiang Zeduoji quickly "went out of the circle" with a sci-fi propaganda film and fluent English, becoming another well-known director of online celebrity after Liu Hong.
Up to now, his videos have almost "zero bad reviews", but he is still worried about his high exposure. "My friend said I was ‘ Seek your own way out ’ Exposed to the spotlight, all the subtle movements will be artificially amplified. If the traffic is too large, there may be gossip. " Denzawa Dorje said frankly.
In this regard, Ouyang Min believes that it is not unexpected that the traffic will bite back under the strong secondary creation of short videos, and the directors should be prepared.
Chen Bo, a professor at the National Institute of Cultural Development of Wuhan University, pointed out: "Director Xie ‘ Ugly out of the circle ’ It has indeed enhanced the visibility of the city. From the results, this is a relatively successful marketing. But this is not a good medicine for developing tourism. After the track of the director is saturated, what do the directors in other places rely on to get traffic? "
Chen Bo believes that in order to avoid the traffic itself, directors should clearly realize that publicity is only a means, not an end. Compared with "packaging is new", the cultural tourism industry should take the route of "content is king" and retain tourists with high-quality cultural tourism experience, otherwise it will be short-lived and will not last long.
How to seek "allowance" in "flow"
As more and more directors join the "online celebrity" tide, sharp-eyed netizens find "clues", and the short videos of cultural tours gradually appear homogenization phenomenon — — The directors are scrambling to adopt similar practices such as "cross-dressing show" and "showing beautiful scenery", which makes the public feel aesthetic fatigue.
"Imitation is the least controversial and the least investment." A director of online celebrity Cultural Tourism Bureau with a large number of fans bluntly said that because there is no professional operation team, creativity is easy to dry up and operation is difficult. In order to keep the account updated, we have to follow suit.

Tourists visit Nalati tourist scenic spot in Xinjiang. Xinhua news agency
Not long ago, the barbecue in Zibo was "out of the circle", which also provided a new inspiration for Director online celebrity — — You can’t just "roll in" endlessly in the video, but you should pay more attention to the travel experience.
Mitsuzawa Dorje once held a special meeting to re-examine Zibo’s out-of-circle password, and finally came to the conclusion that — — A city should have a core of fireworks.
"Barbecue seems to be a carrier of the taste of street life, but the affinity and interactivity reflected in this process are the core competitiveness." Yasuzawa Dorje believes that the development of cultural tourism should not only have the "face" of video promotion, but also have the "lizi" of tourism experience.
Some directors of cultural tourism said in interviews that the root cause of the fire in Zibo was to identify the uniqueness of the city. We should not simply imitate it, but give full play to local strengths according to local conditions and create a unique and grounded cultural tourism environment.
"I have been busy attracting investment recently." Online celebrity’s influence is being "realized". "Recently, four companies came to me by name to talk about investment and investment in homestay projects and amusement facilities."
In Dafang County, Guizhou Province, Wu Jiaojiao, deputy director of the online celebrity Municipal Bureau of Culture and Broadcasting, experienced the network dividend — — Online data is converted into offline income. A few years ago, they encountered the problem of "pain point". Some fans bluntly told her that the tourism format of young consumer groups was still in a blank, and there was "something to watch" but not "play" for young people. How to solve it once became a difficult problem.
This year, the local reference big data "suits the right medicine": a variety of new tourism formats have been launched — — Yi people’s immersive script tour, dressing tour, terrace light show, theme performance in the 1980 s and other ingenious activities. "On May 1 this year, the number of young tourists who come generously has also increased. The daily average number of marchers is as high as tens of thousands. Among them, among the leisure tourism consumer groups, young users and parent-child groups after the 90 s accounted for nearly 90%. " Wu Hao said.

Wu Jiaojiao (first from right), deputy director of the Bureau of Culture and Broadcasting in Dafang County, Guizhou Province, is at work. Bright picture
Recently, many directors of online celebrity Cultural Tourism Bureau have been thinking — — How to realize the transformation from "online celebrity" to "long red" and from "flow" to "reserve"?
Combining with relevant research, Ouyang Min gave suggestions to the directors: "As a tourist destination known for its natural scenery, it is suggested to build a selling point to relieve urban pressure, embrace nature, purify the soul and strengthen emotional experience. Tourist destinations with profound cultural heritage can focus on the development of folk resources, such as traditional crafts, folklore, historical allusions, etc., and explore deeper cultural connotations. "
A director of the online celebrity Cultural Tourism Bureau said that in the face of the cultural tourism fever in various places, it is also necessary to be wary of rushing into the project.
In this regard, Chen Bo, an expert who has been paying attention to cultural studies for a long time, reminded that the local cultural tourism departments should also do a good job in evaluating the online traffic conversion ability on the premise of clearly publicizing and positioning. "After all, online traffic cannot be directly realized. The cultural tourism department should measure the return on investment of the offline tourism industry and adjust the cultural tourism development strategy in real time according to the evaluation results." Chen Bo said.
Pork prices fell for the first time in 19 months! There are also eggs and duck meat in the price reduction! What signal?
The national consumer price index CPI in October released by the National Bureau of Statistics today rose by 0.5% year-on-year and decreased by 0.3% quarter-on-quarter. Food prices are still the core factor affecting the downward trend of prices. PPI, the ex-factory price index of industrial producers, remained stable year-on-year and quarter-on-quarter.

From a year-on-year perspective, CPI rose by 0.5%, and the growth rate dropped by 1.2 percentage points from last month. Mainly due to the high comparison base in the same period last year, the reduction of hikes and the impact of pork prices from rising to falling.
Specifically, pork prices fell for the first time after rising for 19 consecutive months, down 2.8% year-on-year; However, the year-on-year prices of eggs, chicken and duck are in the negative growth range, and the decline has expanded. The prices of eggs, chicken and duck decreased by 18.3%, 13.2% and 6.5%, respectively, with a decrease of 0.6, 4.1 and 1.8 percentage points.
In addition, the year-on-year increase in the prices of fresh vegetables, beef and mutton also declined, which made food prices the core reason for the decline in CPI. Non-food prices remained basically stable, but the prices of gasoline and diesel dropped significantly, with gasoline and diesel prices dropping by 17.4% and 19.1% respectively.

Alvin Wong, Dean of Founder Mid-term Futures Research Institute: Overall, the decline in food prices is a major reason for the decline in CPI; In terms of non-food, some energy prices also fell in October with the changes in the international market. Therefore, CPI continued to be in a downward trend in October.

PPI, another price index, dropped by 2.1% year-on-year, the same as last month, and industrial production prices remained stable as a whole.

Liu Xinwei, senior researcher of Zhuo Chuang Information: Overall, the infrastructure real estate data is better than expected; The price of black products rose month-on-month. With the increase in heating demand in the north, coal prices maintained an upward trend in October, achieving positive growth year-on-year; In terms of crude oil prices, the decline accelerated in late October, or touched the floor price again.
Top Ten Events of China Real Estate in 2016
Housing prices in first-tier cities skyrocketed at the beginning of the year.

The property market in early 2016 is very similar to the stock market in early 2015. Everyone talks about stocks and everyone talks about houses. Housing prices in first-tier cities have gone up like a runaway, and Shanghai has even seen a price jump of 700,000 a day. At the same time, the scene of buying a house is no less than the double eleven.
In Shanghai, Lujin Shanghai School attracted nearly 100 buyers to queue up overnight in the middle of the night before the opening, and even friends in the team came wrapped in quilts, which surprised many people in the industry.
In Hangzhou, the second phase of Yangliu County in Greentown opened, and even 1000 people grabbed the source of 388 suites.
In Guangzhou, Vanke Mountain View City, which was just the opening time before, couldn’t wait to push it after the holiday, and it was robbed half an hour after the opening.
In addition to the bursting of real estate, the real estate trading center is also overcrowded, and Shanghai Baoshan Trading Center has to implement current-limiting measures.
On the one hand, the real estate in first-tier cities is an ideal haven, and safe-haven investment has become the most direct factor to promote this round of skyrocketing. On the other hand, at the beginning of this year, the central bank lowered the down payment ratio, and social funds also participated. The loose credit policy provided financial solutions for buyers, and financial services similar to "down payment loans" also boosted the rise of housing prices to some extent.
De-leverage the property market and strictly control the "down payment loan"
The so-called down payment loan refers to the fact that when the down payment funds of buyers are insufficient, real estate agents or financial institutions can provide them with subsidy funds for borrowing, which can be used by buyers to enlarge the leverage of buying houses. In some areas where house prices are rising rapidly, the existence of down payment loan has reduced the actual down payment of some buyers to 10%, which is equivalent to 10 times the leverage, and the leverage ratio is much higher than the "off-site fund-raising" at the peak of the stock market last year.
With the skyrocketing housing prices in first-tier cities, "down payment loans" began to surface, attracting the attention of the regulatory authorities. Shenzhen was the first to take the shot. In March this year, Shenzhen Financial Office issued a letter to prevent financial risks in the real estate industry, requiring Internet finance associations and small loan associations to investigate the situation of leveraged lending by enterprises under their jurisdiction and sort out the product model and the amount involved. At the same time, Guangzhou also began to investigate P2P and small loan companies to issue down payment loans.
A regulatory storm against leveraged funds represented by "down payment loans" is coming. If house prices continue to rise, using leverage may make a lot of money; Once house prices fall, there may be "trampling". If the high leverage of housing market is allowed to continue, it will be a financial disaster.
Real estate in first-tier cities is overheated, and the government urgently regulates it.
On March 25th, the Shanghai Municipal Government promulgated "Several Opinions on Promoting the Stable and Healthy Development of the Real Estate Market" to strictly control the property market, among which the most stringent provisions include: "Non-registered households in this city must pay social security for five years or more; For households with a set of housing, the down payment ratio for purchasing improved ordinary self-occupied housing has increased to 50%, and the down payment ratio for purchasing non-ordinary housing has increased to 70%. "
This regulation policy is called the strictest regulation in Shanghai. Coincidentally, Shenzhen has also introduced a regulation policy: non-deep households buy houses in Shenzhen, and the social security payment period is increased from the previous year to three years; The first suite paid off the mortgage, and when buying the second suite, the down payment was increased from 30% to 40%.
Under the strict control policy, the transaction volume of housing prices in Shanghai and Shenzhen dropped sharply, followed by the sale of some properties at reduced prices. Different from Shanghai and Shenzhen, due to the crowding-out effect of regulation, the property market in second-tier cities, such as Suzhou and Nanjing, began to boom, and the transaction volume and transaction price rose.
The expiration of some real estate land use years in Wenzhou has aroused the concern of the whole people.
In April this year, some citizens in Wenzhou were unable to conduct normal transactions when buying and selling second-hand houses. The reason was that the land had expired, and they had to pay hundreds of thousands of yuan of land transfer fees to re-apply for land certificates. They spent millions of yuan to buy houses, and they had to pay a lot of money after the expiration. After our house expires, we have to spend a lot of money to renew it, which has aroused heated discussion and concern from all walks of life.
The property right of the house we live in is divided into two parts, one is the house ownership, which is permanent, and the other is the land ownership. The right to use residential land is generally 70 years, which is also a controversial place.
What should I do if the property right expires after 70 years? According to the relevant provisions in the Property Law, the house is "automatically renewed" when it expires, but it has never been said that this is automatically free.
On the morning of December 23rd, Wang Guanghua, Vice Minister of the Ministry of Land and Resources, said that there was no need to apply for renewal, charge no fees, and handle the transaction and registration procedures normally, which was a clear solution. However, Wang Guanghua also mentioned that this is a transitional approach, and the formal approach has to wait for the introduction of relevant legal arrangements.
There are frequent land kings in first-and second-tier cities, and the price of "flour" is higher than that of "bread"
In May this year, the land market in Shanghai ushered in the first land transfer after the regulation of "Shanghai Nine Articles". The three plots of land in Fengxian and Songjiang all had a premium of more than 100%. The floor price of the two plots in Songjiang was close to 40,000/square meter, and the guaranteed price was 60,000/square meter, which was higher than the regional house price. A week later, the Zhou Pu plot was auctioned, with a floor price of 54,500/m2 and a guaranteed price of 79,000/m2. Similarly, in Nanjing, Hangzhou, Xiamen and Hefei, there have been cases of high premium land acquisition.
Developers seem to be crazy about taking land, the main reason is that they are worried about the future land supply. Take Shanghai as an example, Shanghai has locked up 3,226 square kilometers of construction land, and by 2013, 3,070 square kilometers have been used, leaving less than 156 square kilometers (including business and industry) in the future. Therefore, the scarcity of land in Shanghai has reached an unimaginable level. Coupled with the downturn of the real economy, the real estate market has become a safe haven for wealth.
Shanghai exploded rumors of the new mortgage policy, and divorced people crowded into the Civil Affairs Bureau.
At the end of August, a crazy divorce tide was staged in Shanghai. Divorced people crowded the civil affairs bureaus, and some civil affairs bureaus had no choice but to start limiting the number. Shanghai people’s desperate divorce stems from a rumor that "after September, buyers who have been divorced for less than one year will be treated according to their family situation before divorce." As a result, families who were afraid that they could not buy a house after the New Deal began to queue up for divorce. Although, it was later proved that this was a rumor fabricated by a real estate salesman. However, divorce to buy a house has always existed. Marriage is to protect the family, but now it has become a tool for profit-seeking. Is this the irony of the marriage system or a silent accusation against the real estate system?
Real estate in first-and second-tier cities is out of control, and many cities have introduced purchase restriction policies.
In September this year, the property market went crazy again. Nanjing sold 1,500 sets of houses, and Hefei property buyers rushed for houses in the rain. The price of a building more than 20 kilometers away from the main city of Hangzhou jumped directly from 6,000 yuan to 9,500-11,500 yuan. In Shenzhen, a 6-square-meter pigeon cage with a price of 150,000 yuan per square meter broke out, and the people of the whole country were like witchcraft.
With the booming property market, the purchase restriction policy was immediately introduced. Around the National Day Golden Week, the purchase restriction orders of nearly 20 cities were intensively introduced, and the real estate market in many places was sorrowful. The turnover of new houses in hot cities such as Beijing, Hangzhou and Nanjing declined in a cliff-like manner. At the same time, due to the influence of the New Deal and the adjustment of the down payment ratio, many property buyers have to cancel their contracts because they can’t afford to buy a house.
On the other hand, due to the property market restriction, some funds began to switch to A shares or overseas insurance, which objectively created the structural market of A shares after the National Day and the enthusiasm of mainland investors for Hong Kong insurance.
China CDSWill the real estate bubble be good when it strikes?
On September 23rd, China Association of Inter-bank Market Dealers "Business Rules for Pilot Credit Risk Mitigation Tools in Inter-bank Market" (hereinafter referred to as business rules) and related supporting documents, based on the original two products, launched two new products, including credit default swaps (CDS).
In the movie "Big Short", the protagonist from Wall Street saw the bubble of real estate subprime loans and made huge profits by shorting CDS. Therefore, CDS is also considered to be an important driver of the 2008 financial crisis.
Whether the China version of CDS can help squeeze out the real estate bubble remains to be seen.
Li Ka-shing sells mainland property and withdraws from China.
In October this year, Changjiang Industrial Real Estate Co., Ltd., a subsidiary of Li Ka-shing, announced that it would sell the "Century Hui" complex in Lujiazui, Shanghai for 20 billion yuan. This transaction will bring Cheung Kong Real Estate 5.43 billion yuan, which is also the largest property sold in mainland China by Li Ka-shing so far.
At the same time, Li Ka-shing focused its investment on Europe — — Britain, in particular, has invested in power, natural gas, transportation, retail and other fields, and focused on infrastructure. Britain has become Li Ka-shing’s biggest profit source.
The main reason for Li Ka-shing’s selling of mainland real estate is that there are too many bubbles in mainland real estate, and the risks outweigh the opportunities. Early action can make the company in an invincible position.
The Central Economic Work Conference decided that "the house is for living, not for speculation"
In December, the Central Economic Work Conference was held in Beijing, which mentioned that "houses are used for living, not for speculation", which not only curbed the real estate bubble, but also prevented ups and downs; Cities with high pressure of rising housing prices should reasonably increase land supply, and megacities should speed up the relief of some urban functions and promote the development of surrounding small and medium-sized cities; It is necessary to speed up the legislation of the housing leasing market and accelerate the development of institutionalized and large-scale leasing enterprises.
According to the content of real estate in the meeting, it can be inferred that the era of getting rich by real estate speculation has passed and real estate will return to the residential property; The functions of first-tier cities such as Shanghai and Shenzhen will be evacuated, and there will be more opportunities in surrounding small cities; The focus of the real estate industry will gradually shift from the buying and selling market to the leasing market, which may be the next opportunity for the real estate industry.
[See you at 8: 00] The relevant state departments decided to file an investigation on FedEx!
At 8 o’clock every day, CCTV will sort out the big and small things that happened around us within 24 hours.
[concern]
The relevant state departments decided to file an investigation into the alleged damage to the legitimate rights and interests of Chinese users by FedEx.

Recently, FedEx did not deliver the express mail according to the name and address in China, which seriously damaged the legitimate rights and interests of users and violated the relevant laws and regulations of China’s express delivery industry. The relevant state departments decided to file an investigation.
Great joy! Mother-daughter traffickers were sentenced to death, and buyers were also jailed.

On May 31st, the Intermediate People’s Court of Yangquan City, Shanxi Province publicly pronounced the case of 12 people abducting and selling abducted children across provinces. The defendants Wang Xi ‘e and Liu Liping were sentenced to death for crime of trafficking in children.
It was found through trial that since 2015, Shanxi defendants Wang Xi ‘e, Liu Liping and some family members have bought a large number of babies through various channels in order to seek illegal interests, and sent them to Shandong for sale through Shandong defendants Wang Chuanbin, Li Xiushan and Xu Qingquan. Shandong defendants Xu Qingquan, Yan Shaofu and Chen Guangzhou knew that they were trafficked children, but they still bought them themselves or helped their relatives buy them for adoption.
According to the number of abducted children in this case, combined with the nature, circumstances, degree of social harm and guilty attitude of each defendant, Yangquan Intermediate People’s Court sentenced the defendants Wang Xi ‘e and Liu Liping to death, deprived them of political rights for life and confiscated all their personal property. Sentenced the defendant Wang Chuanbin to life imprisonment, deprived of political rights for life, and confiscated all personal property; The remaining defendants were sentenced to fixed-term imprisonment ranging from 15 years to 6 months, and property or fines were also confiscated.
[domestic]
The exhibition center of Beijing Winter Olympics and Paralympic Winter Games is open to primary and secondary school students for the first time.

On June 1st, more than 200 students from Beijing sang songs such as "We are successors of communism" and My People,My Country in front of the exhibition center of Beijing Winter Olympics and Paralympic Winter Games, and the door of the exhibition hall slowly opened with their loud songs. Under the guidance of the commentator, the children learn about the progress of the preparations for the Beijing Winter Olympics, learn Olympic culture and knowledge, and be familiar with the Winter Olympics and Paralympic Games.
For the first time, BOCOG opened the exhibition center of Beijing Winter Olympics and Paralympic Winter Games to primary and secondary school students. From June 1st, primary and middle school students in China can visit the exhibition center in an organized way through the education administrative department.
Investigation on 108 Black and Evil "Umbrella" Cases in Zhaotong, Yunnan Province
According to the website of the Yunnan Provincial Commission for Discipline Inspection and Supervision, up to now, Zhaotong City, Yunnan Province has filed an investigation on 108 people suspected of being involved in black, evil and corruption and acting as "protective umbrellas" for evil forces. 53 people have been dealt with, including 27 people who have been punished by party discipline and government affairs, and 26 people who have been dealt with by organization and accountability; Fifteen people were detained, and four people were transferred to judicial organs for review and prosecution according to law.
Forbes China list of scientific and technological women: Tu Youyou and other 50 people were selected.

Forbes published the "China list of scientific and technological women" for the first time, and 50 women including Tu Youyou, lifelong researcher and chief researcher of Institute of Traditional Chinese Medicine, Chinese Academy of Traditional Chinese Medicine; He Tingbo, president of Huawei Hisilicon; Ge Yue, vice president and managing director of Greater China of Apple; Du Hong, president of Sina /COO; and Liu Qing, president of Didi Chuxing, entered the list. Forbes China said that the list is a ranking-free evaluation of 50 outstanding women in science and technology. They are founders, executives, front-line R&D engineers or pioneers of technological innovation.
Haikou duty-free shop opened in the second phase, with a business area of 22,000 square meters.
The second phase of Haikou Duty Free Shop opened on June 1st. So far, Haikou’s only duty-free shop with a business area of 22,000 square meters has been fully opened. Haikou Duty Free Shop is located in Riyue Square of the city, with a second-phase operation area of 9,000 square meters. At present, the duty-free shops are selling nearly 200 internationally renowned brands of duty-free goods.
[international]
Gun case in the United States: one injured person died, and the number of victims rose to 12.

According to comprehensive reports, a large-scale shooting incident occurred in Virginia, USA. As one injured person died on the way to hospital, the number of victims in this incident rose to 12, and many others were injured. In addition, the gunman also died after exchanging fire with the police.
The identity of the seven victims of the Hungarian shipwreck was confirmed. The captain denied the violation.

A tourist boat collided with a cruise ship in the Danube waters of Budapest, Hungary, killing seven Koreans on board. Recently, the South Korean Foreign Ministry said that the identity of the victims has been confirmed. In addition, the captain of the cruise ship was arrested, but he denied breaking any regulations or breaking the law.
The new zip line project of Eiffel Tower is hard for tourists to experience.

According to British media reports, a zip line with a height of 377 feet (about 115 meters) and a length of 2,600 feet (about 792 meters) has been built on the Eiffel Tower, a landmark building in Paris, France, for visitors to experience the thrill of crossing the sky quickly. It is understood that the project is "one ticket is hard to find": those who want to participate need to register on the homepage of brand social media, and the lucky ones are randomly selected.
Society
Tricycle stole 1 ton of stone lion. Thieves and cultural relics dealers were all detained.
A few days ago, a quadrangle in Dongcheng District of Beijing was stolen, and a stone drum and a stone lion weighing nearly a meal were stolen at the door. After investigation, five men stole the stone drum lion with an electric tricycle and sold it to Liu, a cultural relic dealer. On June 1, Dongcheng police reported that the stolen stone drum was identified as a cultural relic. At present, five men who stole cultural relics were criminally detained according to law on suspicion of stealing cultural relics and collecting stolen goods.
Guangdong police cracked a huge case of false invoicing, involving more than 5.3 billion yuan.
According to the Guangdong Provincial Public Security Bureau, police in Shenzhen, Guangdong Province recently busted a criminal gang that falsely invoiced ordinary VAT invoices, arrested seven people involved and found out more than 900 units that falsely invoiced, involving more than 5.3 billion yuan.
Nanning police quickly handle a hostage-taking case. Police stormed and subdued the suspect.
Xixiangtang Branch of Nanning Public Security Bureau of Guangxi reported on the 1st that on May 31st, the bureau, together with the special police detachment of Nanning Public Security Bureau and other units, quickly handled a sudden case of hostage-taking, arrested the suspect Zhou on the spot, and the detained person Wei was safely rescued. After preliminary investigation, the case was triggered by a family dispute. The public security organs are currently further investigating the case.
Hunan: Arrested 46 telecom fraud suspects, involving millions of yuan.
Recently, the People’s Procuratorate of Furong District of Changsha City approved the arrest of 46 suspects, including Yang Moumou, on suspicion of fraud. At present, this case is under further investigation. Since February 2019, in the name of the company, Yang Moumou and others have organized employees to falsely register female accounts through social software and add netizens. After gaining the trust of the victim, the criminal suspect induced the victim to register and recharge the "five-point lottery" on the Star Road Entertainment website. The criminal suspect defrauded others of money by manipulating the results of the "five-point lottery" in the background, involving millions of yuan.
[Sports]
Ma Long/Wang Chuqin won the runner-up in the men’s doubles final of 2019 China Table Tennis Open.

On June 1st, in the men’s doubles final of the ITTF World Tour 2019 China Table Tennis Open in Shenzhen, China team Malone/Wang Chuqin lost to German team Boer/Francesca 0-3 and won the runner-up.
[face]
[Zhan Guangwei] The teacher "Frozen Man" has been sticking to the podium for more than 20 years.

More than 20 years ago, Zhan Guangwei, who first came to the podium, was diagnosed with "progressive muscular dystrophy", commonly known as "frozen person". With the continuous deterioration of the disease, from being able to walk by yourself, to needing help, and then to sitting in a wheelchair … … Nowadays, he even has a hard time getting textbooks and teaching AIDS. However, this teacher still sticks to the front line of education and continues to "radiate heat" in the classroom.
See you at 8 o’clock tomorrow!
Editor: Zhu Xinyu
Authoritative departments intensively voiced multiple policies to escort and stabilize foreign trade.
"The monthly container production capacity has increased to 500,000 TEUs, and the shortage of empty containers in major ports in China has dropped to 1.3% in May, which has gradually eased." On June 24th, at the press conference held by the State Council Office, foreign trade logistics became one of the hot spots. In fact, tight logistics and rising freight rates have always been difficult problems faced by foreign trade enterprises in the near future, and they are also the focus of the work of relevant departments.
The reporter of the Economic Information Daily noticed that in just two days, at the the State Council executive meeting, the press conference of the State Council Office and the press conference of the Ministry of Commerce, a number of authoritative departments voiced their voices one after another, focusing on strengthening cross-border logistics cooperation, increasing transport capacity guarantee, freight rate supervision, strengthening supply and price stability, and stabilizing exchange rate, and brewing more support policies to help foreign trade enterprises develop and promote the stability and quality of China’s foreign trade.
All parties actively respond to ease the pressure on foreign trade enterprises.
"The capacity of 23 million TEUs is now put into shipping except for about 1% of the ships under repair, and even some non-container ships come to carry containers." At the press conference of the State Council Office on the 24th, Zhao Chongjiu, Vice Minister of the Ministry of Transport, introduced it.
On the same day, Gao Feng, spokesman of the Ministry of Commerce, said that Guangdong Province is taking effective measures to co-ordinate the prevention and control of epidemic situation and smooth the international logistics work, and do its utmost to solve the problem of ships pressing on Hong Kong. From 0: 00 on the 24th, the overall operation of Shenzhen Yantian Port was fully restored.
The reporter saw at the scene of Yantian Port that there are berths in 20 berths in the central and western port areas, and all the operating shore cranes have been fully opened.
These are just the epitome of all parties helping import and export enterprises to solve problems. Since last year, the shortage of international logistics has become a headache for foreign trade enterprises. "Some customers with relatively weak strength can’t bear the cost pressure caused by this, and the goods can’t be shipped out and piled up in the warehouse, which increases the inventory and turnover cycle." Chloe Wang, vice chairman of the board of directors of Shengwei International Holdings (China) Co., Ltd., told the Economic Information Daily.
"This year’s sales have increased a lot compared with the same period of last year, but the profit side is not very optimistic." A person in charge of a foreign trade enterprise in Ningbo said frankly that the increase in freight and raw material prices squeezed the profit margin.
Gao Shiwang, director of the Industry Development Department of the China Chamber of Commerce for Import and Export of Mechanical and Electrical Products, told the Economic Information Daily that these factors have a great impact on foreign trade enterprises, especially some industries where raw materials account for a high proportion of costs, such as household appliances, low-voltage electrical appliances, machinery and tools. The transportation problem increases the inventory cost, and demurrage, container pressing and overnight expenses add up to be a big burden.
Solving these problems requires many efforts. In terms of customs clearance convenience, the General Administration of Customs has recently jointly launched 18 policy measures with the National Development and Reform Commission, the Ministry of Finance, the Ministry of Commerce, the Ministry of Transport, the General Administration of Market Supervision and other departments, focusing on optimizing processes, reducing costs, reducing time and improving efficiency, etc., and making efforts to solve the blocking points, pain points and difficult problems that the current market players are concerned about.
Some measures have achieved initial results. The reporter learned at Nanjing Customs that the customs introduced Yangshan — The new supervision mode of "joint loading and unloading, treating as one port" in Taicang Port realizes one-time declaration, one-time inspection and one-time release of container goods, effectively improving customs clearance efficiency.
In beibu gulf port, through scientific and reasonable organization of production and overall coordination of production resources in the three ports, the shortage of shipping space on ocean routes has been effectively alleviated. From January to May, the container throughput of this port increased by 29.5% year-on-year.
Li Jinhua, Marketing Manager of China Qinzhou Ocean Shipping Agency Co., Ltd., said that customers have been giving full play to the developed domestic transportation system and flexibly choosing delivery points. "Customers in the southwest area may have taken Qinzhou Port more, and now they will take Guangzhou Port or Shanghai Port, which is equivalent to an internal resource optimization and allocation."
Multi-sector intensive deployment, stable foreign trade, and policy strengthening
It is worth noting that stabilizing foreign trade will usher in more favorable policies. The executive meeting in the State Council on 23rd decided to take measures to accelerate the development of new foreign trade formats and new models, promote the upgrading of foreign trade and foster new competitive advantages. The meeting proposed to strengthen international cooperation in the fields of intellectual property protection and transnational logistics.
Multiple deployments are also carried out at the ministerial level. On the 24th, Gao Feng said that the Ministry of Commerce, together with the People’s Bank of China and the State Administration of Foreign Exchange, promoted financial institutions to continuously optimize exchange rate hedging products and services, and actively promoted the Foreign Trade and Economic Cooperation Enterprise Exchange Rate Hedging Business Manual to provide targeted guidance for enterprises to hedge. He also made it clear that he will continue to pay attention to the exchange rate fluctuations and help foreign trade enterprises actively respond.
Earlier, in response to a reporter’s question from the Economic Information Daily, Gao Feng said that recently, the Ministry of Commerce, together with relevant departments, conducted in-depth research on 7,500 foreign trade enterprises through the foreign trade and foreign investment platform, and took measures together with relevant departments and local institutions, such as promoting import diversification and building stable trade channels for bulk commodities. In the next step, we will further optimize and improve the trade policy toolbox to help foreign trade enterprises reduce costs and expand the market.
The Ministry of Transport has made it clear that it will work with the Ministry of Commerce and other departments to continue to implement relevant measures to stabilize foreign trade, guide international liner companies to continuously increase the supply of Chinese mainland’s export routes, improve the container turnover efficiency, and guide local transportation departments to ensure the stability and smoothness of the international logistics supply chain while doing a good job in epidemic prevention and control. In terms of price, it will cooperate with the market supervision department to strengthen the supervision of maritime port charges and investigate and deal with illegal charges according to law.
The General Administration of Customs has previously stated that the next step will be to fill the "short board" in terms of port collaborative governance, deep cooperation between enterprises and enterprises, and cross-departmental data sharing.
Business associations are also actively taking measures. Gao Shiwang said that with the support of the Ministry of Commerce, various import and export chambers of commerce have strengthened communication and cooperation with shipowners’ associations and China Shipping Remote Control, and are seeking more effective solutions for foreign trade enterprises.
Seize the opportunity of development and accelerate the transformation and upgrading
Regarding the situation faced by China’s foreign trade, Pang Chaoran, an associate researcher at the Research Institute of the Ministry of Commerce, believes that the recent recovery of external demand and the return of orders have driven China’s export growth, while short-term unfavorable factors such as exchange rate, inflation, shipping, chip supply and rising labor costs have increased the cost of export enterprises. "But these disturbing factors are exogenous, phased and temporary in general." He told reporters.
Liu Xingguo, a researcher from the Research Department of china enterprise confederation, said in an interview with the Economic Information Daily that at present, China’s foreign trade enterprises not only have to deal with many challenges, but also face greater opportunities, with both favorable and unfavorable factors.
In Pang Chaoran’s view, relevant departments and foreign trade enterprises should seize the opportunity to accelerate the process of moving towards the high-end link of the industrial chain and continuously enhance their foreign trade competitiveness in the process of continuous transformation and upgrading of domestic production capacity.
Pang Chaoran suggested that from the policy point of view, all departments should still pay close attention to the changes in the situation, find out the emerging and trend problems in time, make pre-judgment and early warning, and provide stable policy support for the sustained and healthy development of foreign trade enterprises; Enterprises should accelerate transformation and upgrading, constantly improve the international competitiveness and bargaining power of products, promote short orders to become long orders, and prevent price changes from affecting profits.
Liu Xingguo believes that while government departments take appropriate measures, enterprises should also take the initiative to seize the opportunity to accelerate development: First, strengthen cost management and maximize the pressure of cost increase; Second, scientifically and cautiously use innovative tools in international financial markets to partially eliminate the impact of RMB exchange rate fluctuations; The third is to speed up innovation and development, transformation and upgrading, and enhance the bargaining power of foreign trade enterprises in international trade with high-quality products.
In fact, foreign trade enterprises are already taking active actions. Chloe Wang said that overall, the export market is expanding this year, and the company is further expanding its production capacity and intensifying supply chain integration. "In order to cope with the uncertainty in the future, the company will improve product competitiveness and expand more international markets as an important task in the second half of the year."
Looking at the city from the "portal", where is the emerging power of Fuzhou’s regional rise?












The Spring Festival is different for grassroots researchers, inspiring the spirit of dragon and horse and sticking to the front line of scientific research
Core tips
Some of them insist on racing against time in the laboratory, some insist on answering questions on online science popularization, and some go all out to protect people’s health … This Spring Festival, many researchers are still fighting in their respective posts in different ways. Their figure is a vivid footnote that Henan is playing the "strongest sound" of scientific and technological innovation, and it is also a microcosm of the struggle of all walks of life and all fronts to inspire the spirit of Dragon and Horse and create a better future.
Every second counts for a doctor’s heart
At 7 o’clock in the morning, I rushed to the hospital, went to the clinic for a first-line inspection, then got into the laboratory to discuss the experimental progress with my colleagues … After the work plan for one day was completed, there were thousands of lights outside the window.
This is the daily life of Yu Zujiang, vice president of the First Affiliated Hospital of Zhengzhou University and director of the genetic testing center of the hospital during the Spring Festival holiday.
"There is little difference between the Spring Festival and peacetime, because the disease is not’ holiday’, and our medical workers have to race against time." Yu Zujiang said.
In Yu Zujiang’s eyes, this year’s Spring Festival can already be described as "relaxed". Although he is at work most of the time, he still has time to eat with his family. Previously, as a member of the expert group on epidemic prevention and control in COVID-19, he was busy in epidemic prevention and control for several consecutive Spring Festival.
At 10: 00 am on February 16th, at the Sub-center of the National Key Laboratory of Innovative Drugs for Antiviral Infectious Diseases (hereinafter referred to as the Sub-center) located in the East Campus of the First Affiliated Hospital of Zhengzhou University, when the reporter saw Yu Zujiang, he was discussing the experimental progress with two colleagues enthusiastically.
On the electronic screen of the sub-center, the word "Happy New Year 2024" in gold on a red background reveals a strong flavor of the year. During the Spring Festival holiday, Yu Zujiang’s team has people on duty here every day.
"Before the Spring Festival, we found four new anti-tumor drug targets, and during the Spring Festival, we screened them in the molecular drug library." Yu Zujiang introduced.
"Because drug screening and verification and cell culture are a continuous process, we can’t stop during the Spring Festival." Yu Zujiang said that they are conducting "life-saving" research. If they can produce results early, they can help more patients find hope for life as soon as possible.
As an expert in the field of infectious diseases, Yu Zu Jiangdu has been "fighting" with liver diseases for 20 years.
Previously, Yu Zujiang led the team to study hard for nearly 10 years, and found a drug named trimetazidine, which can become a new target drug for treating liver diseases, and successfully completed the patent transformation with a transfer amount of 2.34 million US dollars.
At present, the new clinical indication of trimetazidine in the treatment of chronic and acute liver failure has applied for an international invention patent and obtained the qualification certification of "orphan drug" (rare drug) from FDA of the United States.
"At present, the experiment being carried out in our laboratory is to find more new drugs for cancer patients." Yu Zujiang said that although these experiments need to go through numerous "trial and error" and may not break through for a long time, as long as there is a glimmer of hope, they will go all out and explore without stopping. (Reporter Li Xiaomin)
Online classes often cultivate "golden" flowers.
During the Spring Festival holiday, the WeChat group of Professor Li Yongchao, the chief science popularization expert of the province and the head of the provincial Chinese herbal medicine industry science and technology correspondent service group (Fengqiu), was particularly lively:
"Teacher Li, my family’s heavy-haired flowers have been planted for seven or eight years. What should I pay attention to when pruning?"
"Thank you, Mr. Li. Every year, we quarrel about cutting two branches of flowers. Last time you came to class and said you couldn’t cut them again, he finally listened this year."
Most of the 267 friends added by Li Yong’s ultramicro letter "Fengqiu Honeysuckle Development Group" are growers. It is a compulsory course for him to reply and answer questions every day during the Spring Festival.
"The temperature in winter is below 0℃, and the plant nutrient solution does not flow out after honeysuckle pruning, so the pruning effect is the best around beginning of spring." On February 17, Li Yongchao told reporters. On the 28th day of the twelfth lunar month, he also went to fengqiu county with members of the service group to give special training on pruning techniques to the technical backbone of local growers.
As an academic and technical leader in the scientific and technological research of flower Chinese herbal medicines and the promotion and service of agricultural science and technology in our province, Li Yongchao has been giving technical guidance in fengqiu county since 2012.
"The golden flowers are silvery, and the vines are clustered. Fengqiu county is the best suitable area for honeysuckle, and the cultivated honeysuckle has good appearance and excellent quality, and the price per catty is one or twenty yuan more expensive than that in other regions. " Li Yongchao introduced that honeysuckle is a real "rich flower".
"The honeysuckle will not enter the full flowering period until the fourth year. According to the technology taught by Teacher Li, I not only took care of Ben in the first three years, but also made a slight surplus. " Cui Jiuhong, a grower in Yingju Town, said excitedly that the 10 mu of honeysuckle he planted now can earn more than 100,000 yuan a year. "The neighbors still use the old method, and their growth is incomparable with mine."
"Smell the flowers and see the industry." As the authentic hometown of honeysuckle in China, the planting area of honeysuckle in fengqiu county is stable at 100,000 mu at present. Since the service group of science and technology commissioners came to Fengqiu in 2018 to carry out technical guidance and training, the technical radiation area in the county has reached more than 5,000 mu, and it is growing at an annual promotion rate of 2,000 mu.
"Everyone can see the effect of advanced technology, so the enthusiasm is particularly high. Now almost every other week, we will go to Fengqiu for on-site guidance. " Li Yongchao said, "We should pay attention to the prevention of aphids in March. I just informed everyone in the group that we will open another training class before the fifteenth day of the first month." (Reporter Yin Jiangyong)
Scientific research "fireworks" are both happy.
On the gate, the red Spring Festival couplets convey the joy and hope of the Spring Festival. In the study, quiet meditation and heated online discussion "record" the persistence and dedication of researchers.
Yu Chen, vice president of zhengzhou institute of aeronautical industry management Institute of Electronic Information, kept switching between "fireworks" and "scientific research".
"Chinese New Year, you must take time to spend more time with your family and bring more children. However, scientific research work is daily for us, and holidays can’t fall. " On February 16th, Yu Chen told reporters that the team teachers were not idle during the Spring Festival.
Zhengzhou institute of aeronautical industry management is speeding up the construction of Zhengzhou University of Aeronautics and Astronautics. The School of Electronics and Information has put forward the plan of "standing on top of the world" to make up for the shortcomings of the university’s construction of aeronautics and astronautics, conform to the development direction of the national "aerospace" strategy, and combine the automobile and aerospace construction plan of our province’s "14 th Five-Year Plan" to carry out technical research and strengthen organized scientific research aiming at key issues.
Scientific research is a protracted war, and it is always a race against time.
As the head of the unmanned platform team in the air and space, when talking about the scientific research during the Spring Festival, Yu Chen said more: "We mainly have four research directions. One is’ Dingtian’, actively integrating into the aerospace industry chain, facing the commercial needs of the ground TT&C operation and maintenance business of satellite launch and long-term on-orbit operation, and carrying out innovative research and development of the baseband equipment software and hardware system of the vehicle-mounted satellite ground station integrating TT&C, data transmission, arrow and remote control; The second is’ site’, which is deeply integrated into the unmanned vehicle industry chain. Facing the problems of unmanned vehicle automatic driving and comprehensive planning and scheduling, the unmanned vehicle terminal is developed, and a big data platform for comprehensive planning and scheduling is built based on multi-source fusion positioning, vehicle-road coordination and environmental awareness; Third, to solve the problem of "data island" of manufacturing enterprises, build a collaborative manufacturing platform of data space and network, realize the whole process integration value chain collaboration and the whole cycle data interconnection; The fourth is to continue to carry out research and development of software and hardware systems for autonomous flying drones, and solve the problem of over-the-horizon autonomous flight and optimal allocation of multi-objective tasks for multi-UAVs. "
The boring technical terms in the eyes of ordinary people are extremely "cute" in the eyes of scientific researchers. Although it is hard to explore the unknown scientific research work, they find Yu Chen more interesting.
"A year’s plan lies in spring. In the new year, we will continue to deepen our efforts in the fields of aerospace technology such as UAV navigation guidance and control, key technologies of satellite ground stations, networked unmanned vehicle terminals and big data platforms. " Yu Chen said with a smile, in fact, every fruitful day is a festival worth celebrating for researchers. (Reporter Shi Xiaoqi)
The flag of spring stands up
During the Spring Festival holiday, Zhang Weixian, director of the Academic Committee of Henan Institute of Animal Husbandry and Economics, rarely spent the next year with his family in Zhengzhou.
But he’s not really idle. Jiaozuo, Zhoukou, Xinyang … Every day, many farmers from all over the province call for advice-what should I do if the broiler market keeps fluctuating? Do you want to adjust the breeding varieties? Is there any room for feed cost to fall?
As an animal husbandry expert, Zhang Weixian patiently analyzed and answered questions for them, while incorporating the feedback from farmers from the production line into his own research.
"The market situation is often affected by many factors. What we scientists and technicians should consider is how to promote the high-quality development of animal husbandry through scientific and technological innovation." In Zhang Weixian’s view, the current problems of animal husbandry boil down to two major aspects, one is germplasm resources, and the other is feed.
In recent years, feed prices have continued to rise and feeding costs have been increasing. Is there a cheap "flat substitute" for imported feed from protein? Zhang Weixian aimed at the research direction of crop straw.
"On the one hand, many straw resources are treated as waste; On the other hand, the shortage of forage resources in animal husbandry has caused the rising cost of breeding, which is simply’ begging for food with a golden rice bowl’. " In order to recycle the straws of these crops, he served as the director of the Key Laboratory of Innovation and Utilization of Unconventional Feed Resources in Henan Province, and led the team to carry out research on the efficient conversion of crop straws into high-quality feed for livestock and poultry, such as peppers, peanuts and sweet potatoes.
"Taking peppers as an example, the planting area of peppers in Henan is about 3 million mu, ranking second in the country, and it can produce about 30 million tons of green pepper stalks every year. We conducted experiments on feeding herbivorous livestock through processing methods such as crushing, biological fermentation, silage and yellow storage, and obtained reliable scientific data. " Zhang Weixian introduced that pepper straw contains 4.1% essential amino acids and is rich in nutrients such as carotene and vitamin C. It is a feed resource with great application prospects, and its economic value after being processed into high-quality feed is even higher than that of pepper itself.
During the Spring Festival this year, Zhang Weixian also received two papers from top international journals, which can be published after revision. At the age of 63, like his own students, he kept writing in front of the computer every day, demonstrating the team’s method of analyzing the nutritional value of feed with polymer spectroscopy.
"I learned a lot of new words from students. When spring comes, I also set a flag. I hope our research methods can form a subversive innovation and provide strong support for the development of animal husbandry." He said. (Reporter Shi Zhe)
Holiday scientific research "does not close"
On the morning of February 17, the traffic on the University Road outside the south campus of Zhengzhou University was woven and full of flavor. Shan Chongxin, vice president of Zhengzhou University, walked through the quiet campus and came to the office of the Key Laboratory of Physical Materials, Ministry of Education, and started a day’s work.
Just after the Spring Festival, Shan Chongxin has already started the "working mode" on the third day of the New Year’s Eve. "Scientific research needs to be focused, and you can concentrate on doing something when you go to the office." He said with a smile.
The laboratory is not deserted. Half of the members of the diamond photoelectric materials and devices team led by Shan Chongxin have been "returned" one after another. Holiday research "does not close". Dong Lin, a member of the team and a professor at the School of Physics of Zhengzhou University, said: "As soon as I have time at home, I log on to the school computer remotely and look at the materials and documents. I came to the laboratory in the third grade."
Indeed, for many years, Shan Chongxin and his team members have almost no concept of weekends and holidays. They are always with diamonds, take the laboratory as their home, take root in the field of diamond photoelectric materials and devices, and devote themselves to scientific research and make unremitting efforts.
Predicted the Rabi oscillation of nitrogen vacancy color center in diamond without microwave excitation, which was confirmed by international peer experiments. High-quality diamond and high-purity diamond Raman crystal were synthesized, and 2-inch optical diamond crystal was developed … Just because of persistent research, the team made a series of heavy innovation breakthroughs in just a few years, which made Henan diamond "shine in the world".
"In the process of scientific research and innovation, any lag may lead to the loss of opportunities. We must continue to tackle key problems with a sense of urgency and speed up the seizure of the commanding heights of science and technology." At the annual summary meeting a few days before the Spring Festival, Shan Chongxin encouraged the team to say.
After the summary meeting, Shan Chongxin and everyone were busy until the New Year’s Eve, and then they put down their work and rushed back to their hometown in Changyuan to reunite with their loved ones for the New Year.
Attachment to his hometown, reluctance to give up his hometown, and wanting to do something for his hometown are also the feelings of Shan Chongxin when he chose to return to Henan nine years ago. "Now Henan attaches more importance to scientific and technological innovation than ever before, and has built a platform for scientific researchers to start businesses. Diamond is a dominant industry in Henan, and it is very happy to be able to do something for the country with what I have learned as my hometown." Shan Chongxin said.
At present, Shan Chongxin’s team is aiming at the forefront of diamond materials and devices development in China, focusing on the key scientific problems of high-quality diamond synthesis and application, and climbing to a higher scientific peak. "It is our direction and goal to break through the technical bottleneck of diamond application in semiconductor, electricity, optics, heat, quantum sensing and other fields." Shan Chongxin said.
In the evening, Shan Chongxin walked out of the office and looked at the stars above the city. He firmly believed that the diamonds in Henan would certainly shine more brightly in the future. (Reporter Ping Cao)
Illustration/Wang Weibin
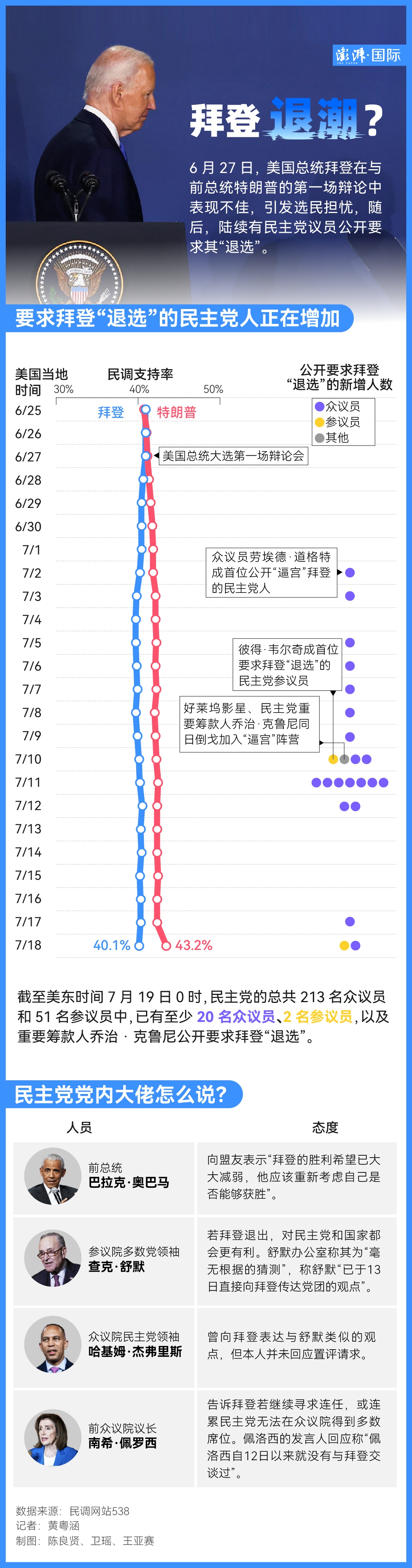
"Nightmare for Three Weeks" before Biden’s Withdrawal: The Spread of Panic and Anxiety and the Increase of "Insiders"
When US Republican presidential candidate Trump welcomed his new running mate in Milwaukee last week, the video clip showed that "the weak Biden struggled to climb the short boarding ladder of Air Force One on his way to further isolation."
This is a bleak scene described in Washington post’s article "Behind the scenes: Biden loses control of the Democratic Party" on July 19th. A person familiar with the matter said that aides forwarded the video in frustration.
According to the report, just four months before the presidential election, Biden’s control of his party suddenly collapsed, which is unprecedented in history. Just a month ago, all these people were United behind Biden, and the mantra of former House Speaker nancy pelosi (Democrat of California) was: "Diversity is our strength. Unity is our strength. "
A source familiar with the situation told Reuters: Biden planned to continue to challenge for re-election as president until last night, but at 1: 45 pm the next day, he suddenly told his senior staff that he decided to withdraw from the election. Biden’s decision caught many White House aides off guard, and several people expressed shock at the news. Because just a few hours ago, Biden’s campaign team was still denying reports that Biden was considering withdrawing from the presidential election.
"The US presidential election has been messed up." A senior consultant who participated in the Democratic Party’s campaign for many years told The Paper that it was hard to hide his disappointment. "You can say anything, predict anything, and draw any conclusions. All in all, this country (election operation mechanism) is out of order. " However, Casey Burghardt, director of the legislative affairs program at George Washington University’s Graduate School of Political Management, recently told The Paper that if properly controlled, the (substitution) procedure can provide an opportunity for the Democratic Party to have a useful debate on its future direction and priorities, and the potential possibility is that it will result in a re-bonded and more strategic party platform.
"Many voters will be ignited by a young candidate who has a better chance of winning." He said, "Almost all Democrats will unite on the first priority: to prevent Trump from winning the second presidential term."
"Make sure he is angry and upset."
As of Saturday (20th) afternoon, 32 Democratic congressmen and four senators have publicly called on Biden not to seek re-election. Among the more than 200 Democrats in the House of Representatives and 51 Democrats in the Senate, this is still a minority. However, Democratic Party officials and party strategists generally believe that the actual number of people who want Biden to withdraw from the election is much higher.
"I’m sure he’s angry and upset." Meghan Hays, who served in the White House until 2022, told NBC that Biden understood the political reality, that is, "he is famous for not holding grudges."
As more and more news revealed that Biden would soon accept the idea of "having to withdraw from the election", The New York Times quoted a person close to Biden as saying on the 19th that in the previous three weeks, the President had insisted that almost nothing could make him withdraw from the election.
According to the report, Mike Donilon, Biden’s long-term senior adviser and one of his most loyal confidants, told him that despite the doubts of the Democratic Party and the pessimistic results of some opinion polls, the competition was still fierce, and he insisted that there was still a chance to win. The president’s family also supported him in sticking it out and pointed out that he had a tradition of overcoming difficulties and not being afraid of questioning.
However, the Democrats behind the scenes and people working in the government described the first three weeks of July as a nightmare-too unusual, too unexpected and unbelievable.
Important members of the presidential campaign team and the White House team privately said that after the disastrous debate on June 27, confidence in Biden began to waver, and the big "money owner" withheld funds and demanded changes.
Then, on a live TV broadcast on July 13th, a more terrible assassination of Trump happened. The former president "magically" escaped the assassination in the attention of the whole world-holding his fist high and his face was still dripping with blood. Biden’s poll remained stable at first, but then began to decline.
Then, when the 81-year-old president went to the election campaign to try to prove his vitality, he was infected with Covid-19 again.
Just this weekend, Biden, who is still isolated from his seaside villa in Delaware because of Covid-19’s infection, got some good news. The former President Clintons, who are still a powerful force in Democratic politics, expressed their support for him privately, and have been actively encouraging donors to support the president, and kept in touch with the White House and offered help within their capacity.
However, former President Clinton and former Secretary of State Hillary Clinton supported their Democratic colleagues to become news themselves, highlighting Biden’s growing isolation in his own party.
A series of actions and policies are hard to turn the tide.
The first week after the debate didn’t go well, and Biden’s team decided to let him "go out" more.
The president participated in three interviews in succession, including the program of Lester Holt, the anchor of NBC Evening News. In the interview, he once showed a combative mood. Although he can’t suddenly look younger, Biden wants to prove his courage and determination to try his best to replace everyone’s image of being old on the night of the debate.
Within 48 hours, Biden also talked with the Congressional Asia Pacific Caucus (CAPAC), the New Democracy Party Alliance, the Congressional Progress Caucus and the Congressional Hispanic Caucus, and won some support.
The White House distributed a list of about 20 other leaders with whom the President talked on the plane or on the trip. About 75 members of Congress reiterated their support for him. "Can you hear the helicopter in the background?" Biden shouted to Reverend Al Al Sharpton on the phone as he was about to board a helicopter to Nevada to win his support. "We must move on, Al."
Sharpton is a famous American black civil rights activist and Biden’s long-term ally. His support is particularly important at all critical moments.
However, a congressman who recently took Air Force One revealed to Washington post that he only recently realized the change of the president after meeting with him many times in the past four years. Biden spoke so softly that he could hardly hear the voice across the table. Another congressman who interacted with Biden recently described him as weak but quick-thinking and seriously considered staying in office.
At the White House, Biden administration officials are also moving quickly. The policy department began to rapidly advance the long-term brewing plan, including drawing up a blueprint for the term limit of the Supreme Court; A legislative proposal has been finalized to set the ceiling of rent increase for business landlords at 5% nationwide in the United States, which may be a large-scale new federal intervention aimed at reversing the main driving factor of inflation.
Politico, an American political news website, reported in mid-July that the aides of Biden’s government had been working behind the scenes all week, showing that the inflation situation finally slowed down through opinion polls and economic data, and appeasing wavering supporters. They hoped that the president’s appearance at the high-profile NATO summit would show his work in strengthening the alliance system.
Members of Congress, allies and presidential aides pay close attention to Biden’s risk points at the press conference. Some of his gaffes provided material for critics, including calling Ukrainian President Zelensky "President Putin" before the press conference, and then correcting and joking about the mistake. Later, he called Vice President Harris "Vice President Trump".
Biden seems to feel the pressure and his temper has become grumpy. The New York Times reported on the 17th that on Saturday (July 13th), Biden lashed out at Representative Jason Crowe of Colorado during a video call with moderate Democrats in the House of Representatives. Crowe, who served in the army and won a medal in Iraq, questioned Biden’s ability to impress voters.
Emotion within the team changed from regret to complaint.
Some people are still cheering, trying to blow the assembly number. "Keep faith." Andrew Bates, senior deputy press secretary of the White House, posted on social media on the 18th.
In the West Wing of the White House (the office of President spin doctor), Washington post’s in-depth report reveals that the loyalty principle that has long accompanied Biden’s actions seems to be disintegrating. According to a Biden administration official familiar with the situation, some consultants turned their attention from planning their second term to planning how to lock in the policy victory "before Trump took office". Other officials said that more efforts are still being put into planning the future agenda.
Among those who are committed to Biden’s campaign, the atmosphere is also deteriorating: they are panicked in private and don’t care about anything.
The crisis seems to be intensifying on all fronts. Democrats are struggling in key States, Biden’s support rate has dropped sharply in all swing States, and long-term donors have warned that Biden’s financial support has dried up.
"The presidential campaign is not over yet. They have no money. " A Democratic Party donation consultant said, "Usually, grassroots funds are expensive and need a lot of (donor) funds to raise these funds, so it is crazy for them to use grassroots funds to make up for the high (donor)."
A senior aide to Biden said that in the campaign team, senior staff were increasingly worried that Biden might lose Virginia. The last time the state supported a Republican presidential candidate was 20 years ago, and now it is usually no longer regarded as a battlefield for presidential elections.
A campaign aide told Washington post that junior and even some senior staff knew nothing about how Biden wanted to advance the campaign. Text messages circulated among Capitol Hill, government officials and others, trying to figure out what happened.
"What’s interesting is that suddenly there seem to be many’ Biden insiders’, whose voices are exactly the same as those of those who blindly quoted and doubted us in successive victories for five years." Bates said.
Some consultants accuse senior assistants of "no longer being loyal, selfish and ambitious" without evidence. A few days later, these slanders even appeared on MSNBC’s "Good Morning Joe" morning news program, which was Biden’s favorite TV talk show.
The New York Times reported that Democrats used to feel sorry for Biden. Although they hoped that he would leave the stage gracefully, now they are increasingly resentful of him, complaining that the president they admired was arrogant and selfish. They feel that he will not only return the White House to Trump, but also drag down other candidates of the Democratic Party.
The call for him to step down is getting louder and louder, not from marginal figures, but from people in the Democratic Party who have been very important to him in the past few years.
The American media found that Biden spent more time with his two longest-serving and most loyal advisers: senior campaign adviser Mike Donilon and presidential adviser Steve Ricchetti. They think their professional knowledge is more popular, and both think that opinion polls have become less meaningful.
The internally monitored poll data comforted the Democratic campaign team. The data shows that most of the supporters Biden lost since the day of the presidential debate are unlikely to vote for Trump, which indicates that they may change their minds.
"I think he bought us some time at the NATO press conference." A campaign consultant revealed to the media that many campaign assistants thought that Biden gave a series of stable and powerful answers after watching the press conference, which eliminated people’s concerns about his health and demonstrated his foreign policy talents. "That night, everyone breathed a sigh of relief. If things don’t go well, I think we all think things will end in 24 hours. "
But the most senior American party political observer can’t deny that no one can force him to submit. "It’s just pure emotion. This is not a strong data-driven action. " A Democratic strategist said that the fate of the Democratic Party depends on the intuition of a person who has been committed to national politics for the past 52 years.




